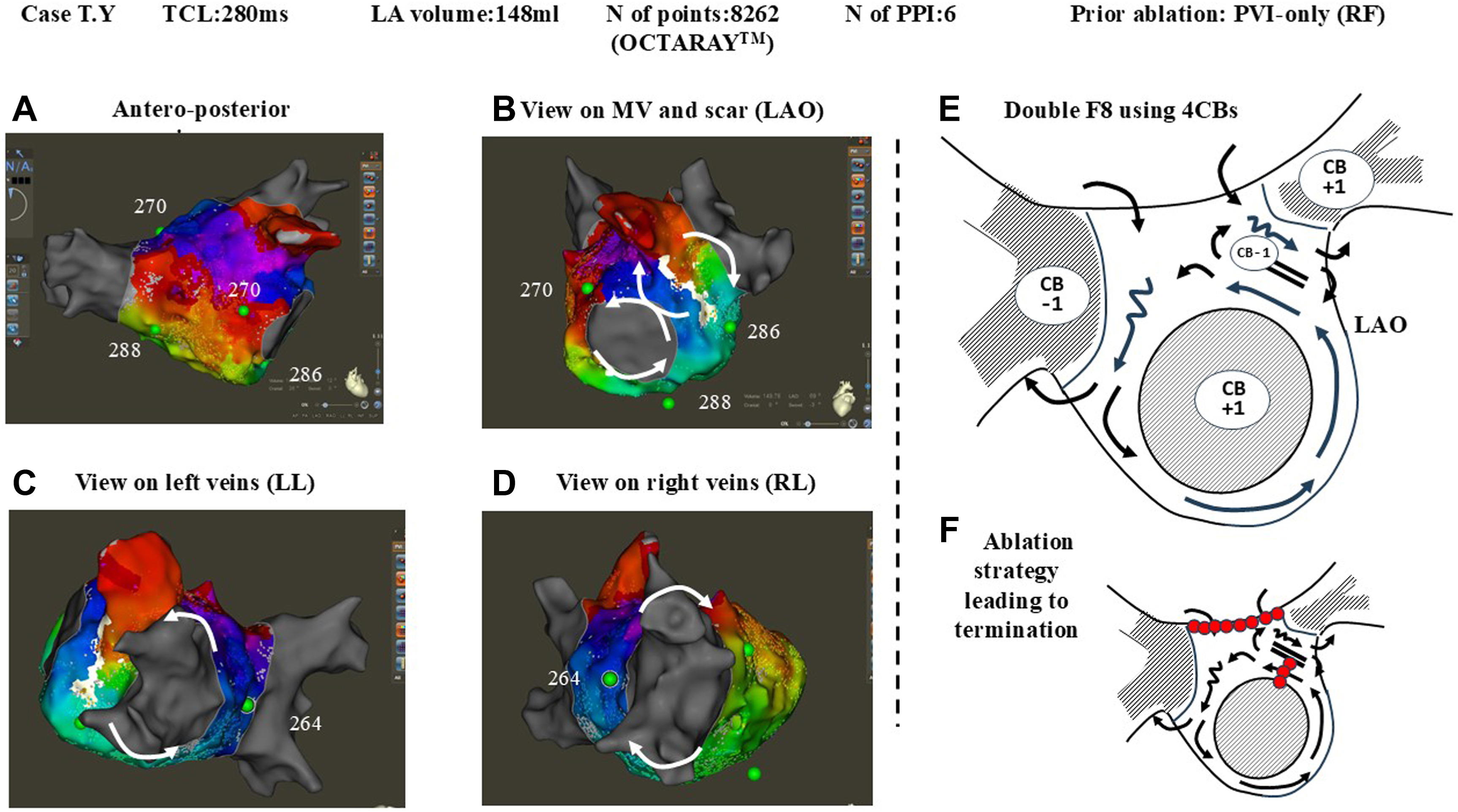
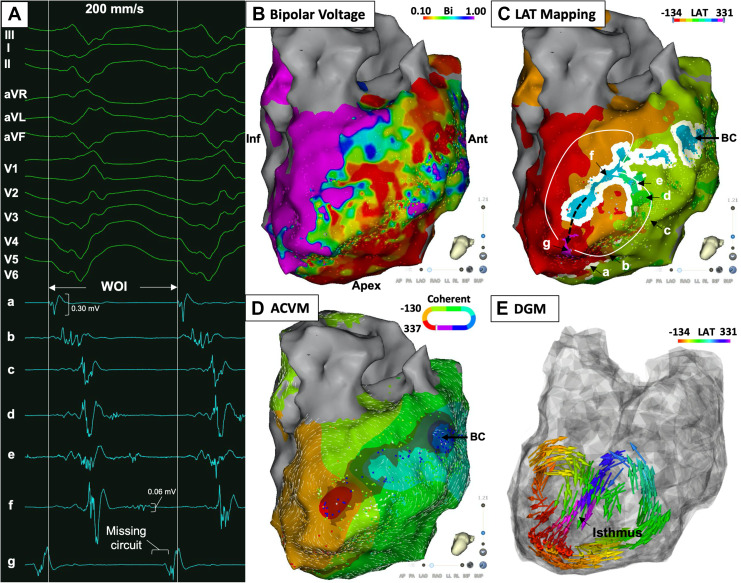
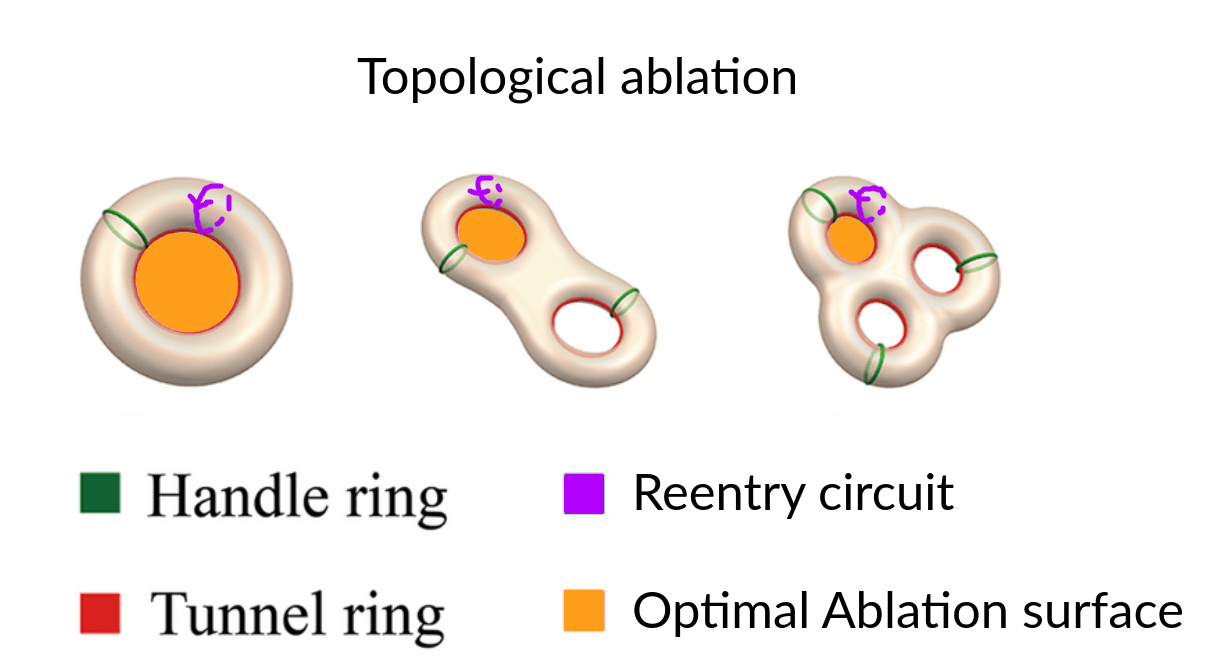
Research
Contents
Publications related to the work explained on this page can be found in the publications section below. Other work, not related to this page, can be found on Prof. Vandersickel's google scholar profile .
1. A unified topological theory of cardiac arrhythmia
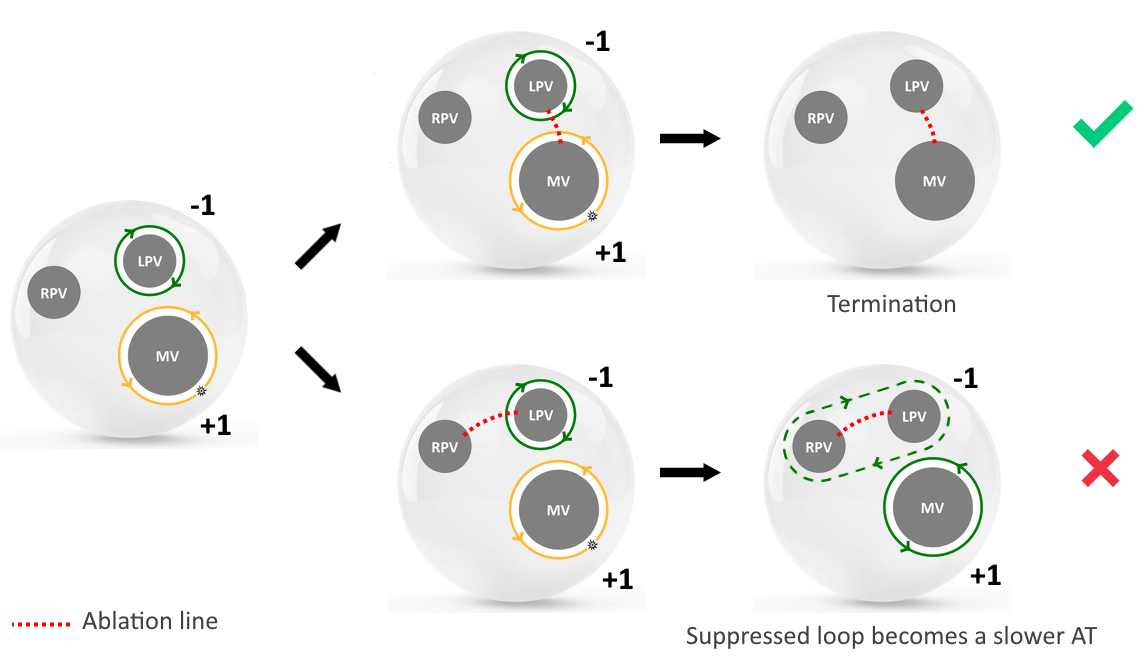
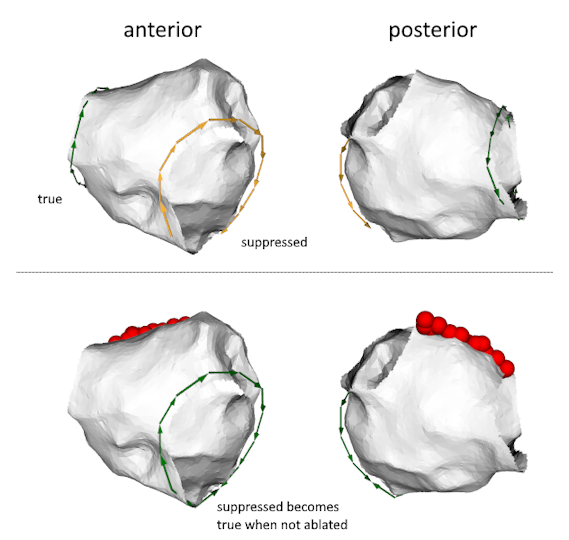
We hypothesize that reentry circuits generally do not exist as isolated entities but usually appear as paired objects: a clockwise reentry is inevitably accompanied by a counterclockwise reentry.
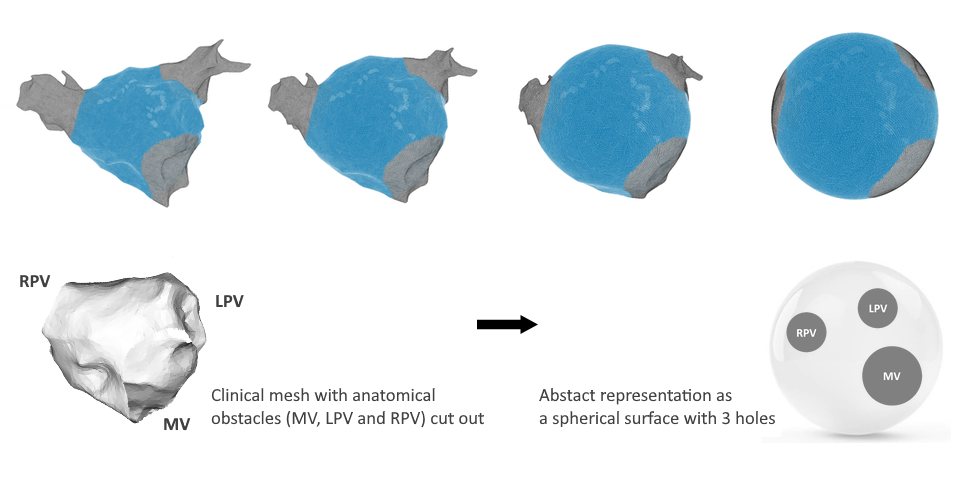
This conceptual breakthrough can be most easily understood by adopting a topological perspective. Topology is a branch of mathematics that studies the properties of objects that are invariant under stretch or deformation. It offers a useful way to simplify the heart’s structure without changing its key features. Topologically, the left atrium (LA) and right atrium (RA) can each be approximated as closed surfaces, equivalent to spheres with a finite number of boundaries10. The left ventricle (LV) and the right ventricle (RV) can also be described as spheres with boundaries, but with finite wall thickness, making their topology considerably more complex 11.
 Topological transformation of a left atria into a sphere
Topological transformation of a left atria into a sphere Topological transformation of a left ventricle into a sphere with thickness
Topological transformation of a left ventricle into a sphere with thicknessA largely forgotten topological theorem, the index theorem for phase singularities. The index theorem for phase singularities, first published in 1977, states that for phase fields defined on closed surfaces with boundaries, the sum of the topological indices must equal zero (Glass, 1977) (Davidsen et al, 2004). The cardiac electrical field serves as a prime example of such a phase field.
As we will show in the next sections, this simple theorem will unify all reentry-driven cardiac arrhythmia.
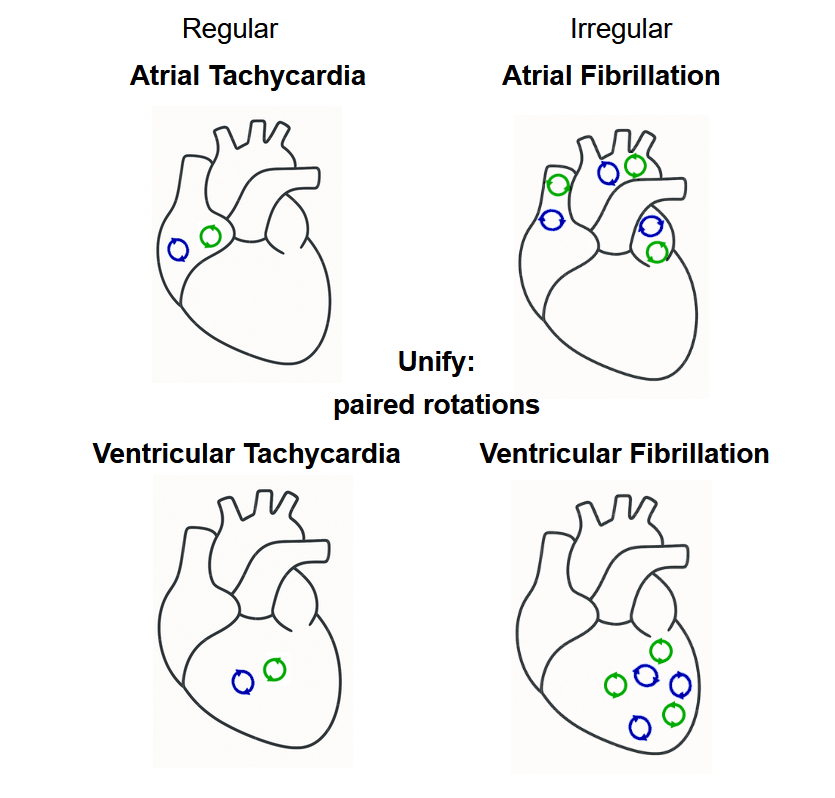 Unification of all major arrhythmia
Unification of all major arrhythmia2. OpenDGM: An open source package to analyze intra-cardiac data.
openDGM2,5 stands as a versatile tool capable of analyzing various arrhythmias, including Atrial Tachycardia (AT), Ventricular Tachycardia (VT), Atrial Fibrillation (AF), Ventricular Fibrillation (VF), and Torsade de Pointes (TdP). It efficiently processes files containing spatial coordinates and simultaneously measured intra-cardiac signals or local activation times (LATs) from electrodes.
While initially developed around network-based analyses, openDGM has evolved into a flexible platform that integrates multiple approaches, including advanced phase mapping, network theory, and topology, for comprehensive dataset analysis. Since becoming open source, we enabled the publication of complete analysis pipelines alongside scientific papers.
No regularity in spatial dataset locations is required, making openDGM independent of the measuring system. It accepts input from computational, experimental (such as needle or socket data), or clinical datasets (CARTO, RHYTHMIA, grid electrode, basket catheter data).
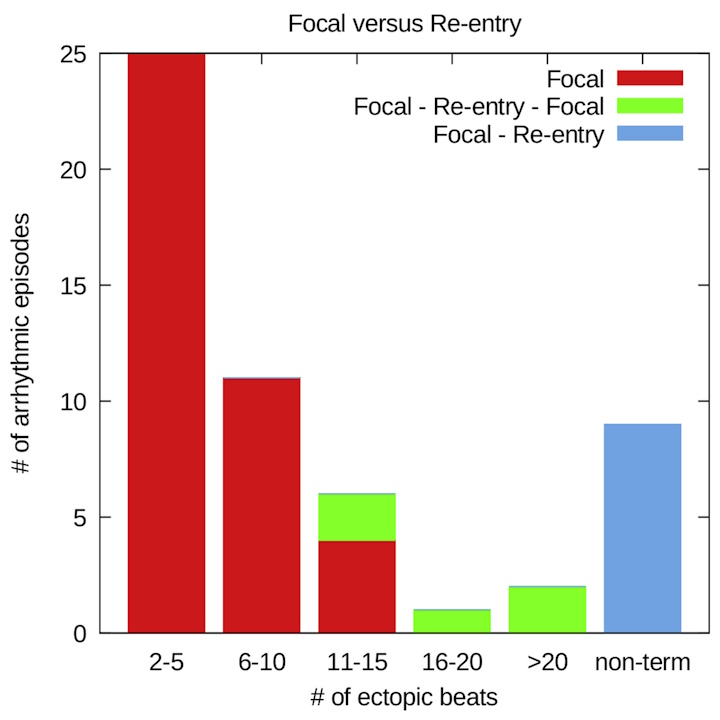
openDGM is recognized for its capability in analyzing reentry loops within arrhythmias, including complete reentry loops, but also incomplete reentry loops. Additionally, DGM can also identify focal sources, although we have not invested yet a large amount of research in this feature.
Mainly, our software package has implemented two main methods: network theory and phase mapping
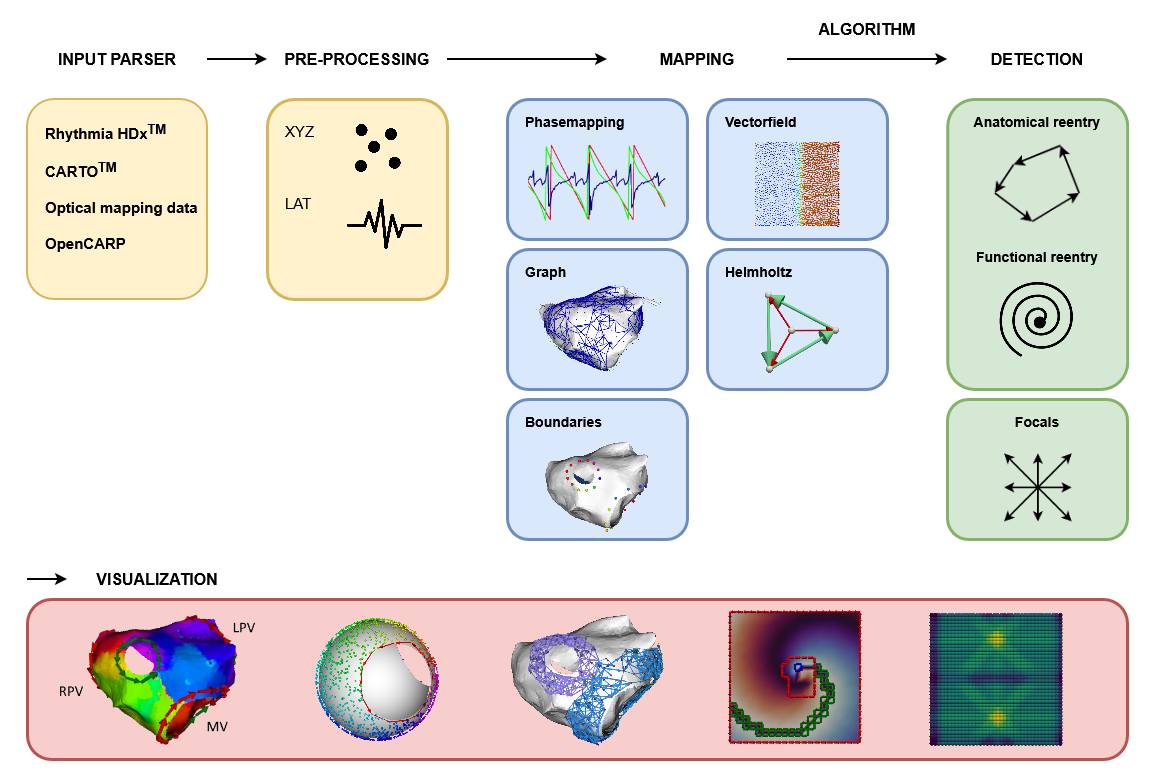 An overview of a complete pipeline for analysis of cardiac activation maps using OpenDGM. The pipeline starts with parsing your desired data format. Then, some preprocessing methods can be applied on this data. Next, mapping will be performed. This can include phase mapping, graph mapping, creating vectorfields etc. Finally, on these maps, our algorithms can detect anatomical and functional re-entry as well as focal sources. To view the result, OpenDGM includes a pyvista wrapper for visualization that works seamlessly with our data output.
An overview of a complete pipeline for analysis of cardiac activation maps using OpenDGM. The pipeline starts with parsing your desired data format. Then, some preprocessing methods can be applied on this data. Next, mapping will be performed. This can include phase mapping, graph mapping, creating vectorfields etc. Finally, on these maps, our algorithms can detect anatomical and functional re-entry as well as focal sources. To view the result, OpenDGM includes a pyvista wrapper for visualization that works seamlessly with our data output.Network Theory
OpenDGM contains a method that transforms electrical mapping data from the heart into a directed network, making it possible to automatically detect the mechanisms driving an arrhythmia, we call this method Directed Graph Mapping (DGM).
DGM takes two simple inputs: the positions of the recording electrodes and their local activation times (LATs): the moment each electrode detects electrical activity. From these, a network is built where each electrode becomes a node, and arrows between neighbouring electrodes are drawn based on the direction and speed of electrical conduction. Only connections with physiologically plausible conduction velocities are included, ensuring the network faithfully represents true cardiac activation.
Once the network is constructed, DGM automatically searches for the arrhythmia source:
- Reentrant activity is detected by finding directed cycles in the network, closed loops of electrical propagation;
- Focal activity is identified by locating regions where all arrows point outward, indicating a local firing site.
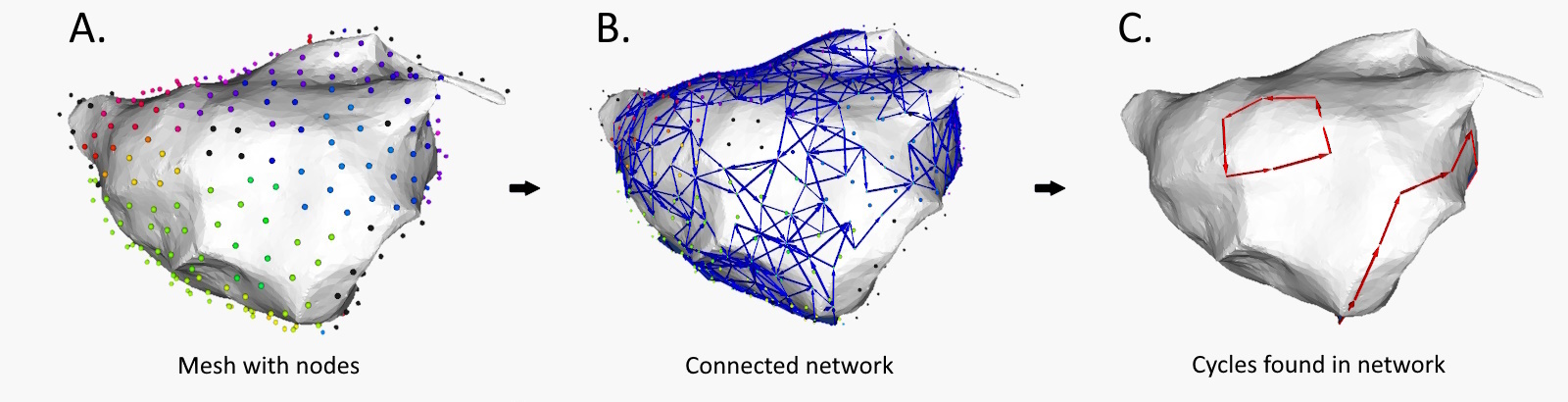 Extracting cycles from the network aligns with the identified reentry loop.
Extracting cycles from the network aligns with the identified reentry loop.Phase Mapping
Phase mapping is one of the most widely used methods for detecting rotational drivers in cardiac arrhythmias. By converting electrical signals into a continuous phase representation, one can identify phase singularities: points around which rotational activity organizes. We have implemented two phase mapping methods in openDGM.
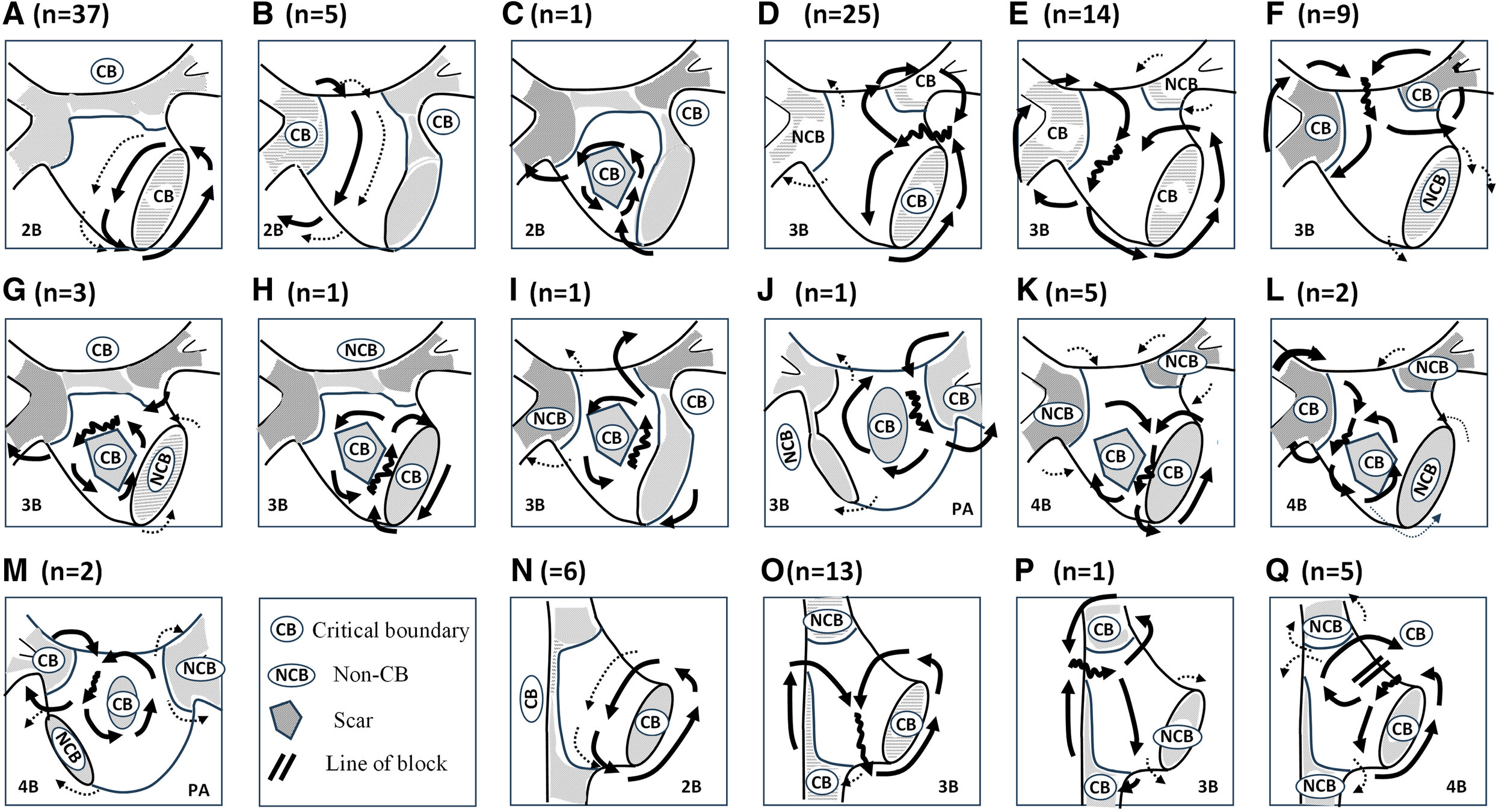
Naive phase mapping is the classical, widely-used approach. It assigns a phase value to each recording point for each time step and identifies rotors by detecting phase singularities. While powerful, this approach has a fundamental assumption: that the phase map is continuous across the entire tissue, except for a finite number of isolated points. In reality, cardiac tissue can contain conduction block, fibrosis, or anatomical boundaries, regions where the phase is either undefined or discontinuous. These regions, called phase defects, cause the naive approach to generate both false positive detections (rotational drivers that aren’t there) and false negatives (rotational drivers that are missed).
Extended phase mapping is an improved implementation developed by our group that explicitly detects and accounts for phase defects 18. Rather than assuming continuity, it identifies where the phase map breaks down, and correctly computes the phase index around these defects. This makes rotor detection robust in realistic tissue, including fibrotic substrates, anatomical boundaries, and clinical data, where the naive approach struggles. Across simulated, experimental, and clinical datasets, the extended approach eliminates erroneous detections and resolves previously missed rotational drivers. This has been overlooked for the past 30 years!
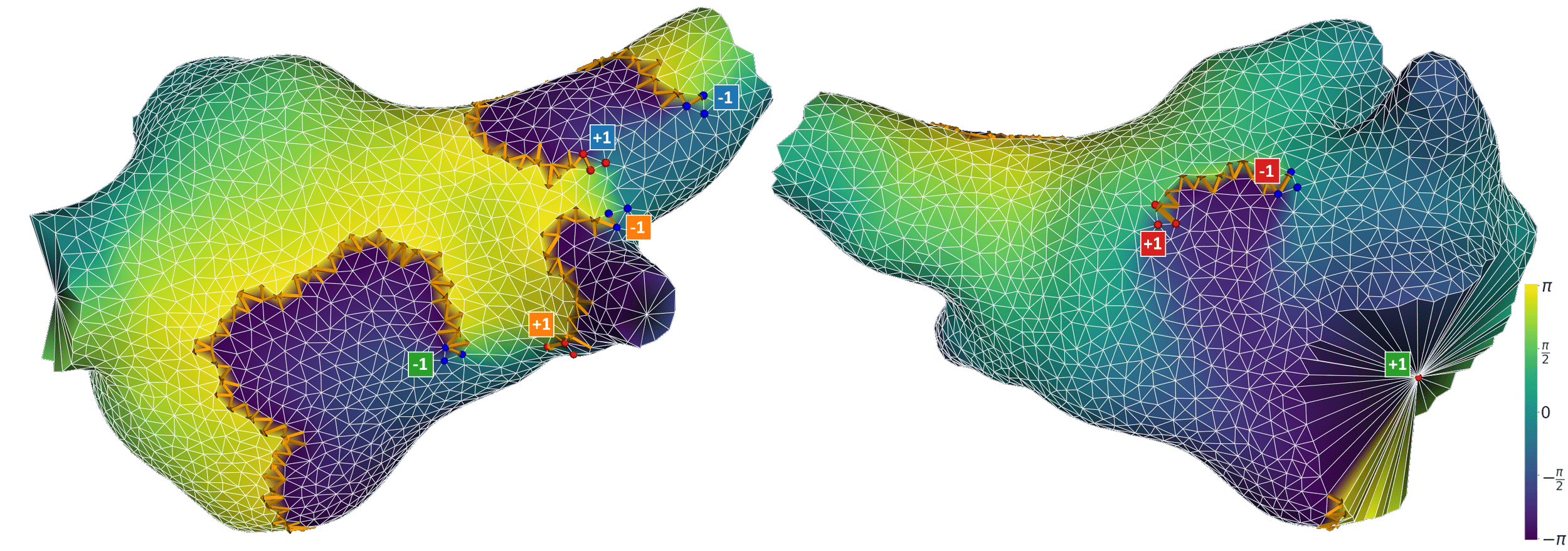
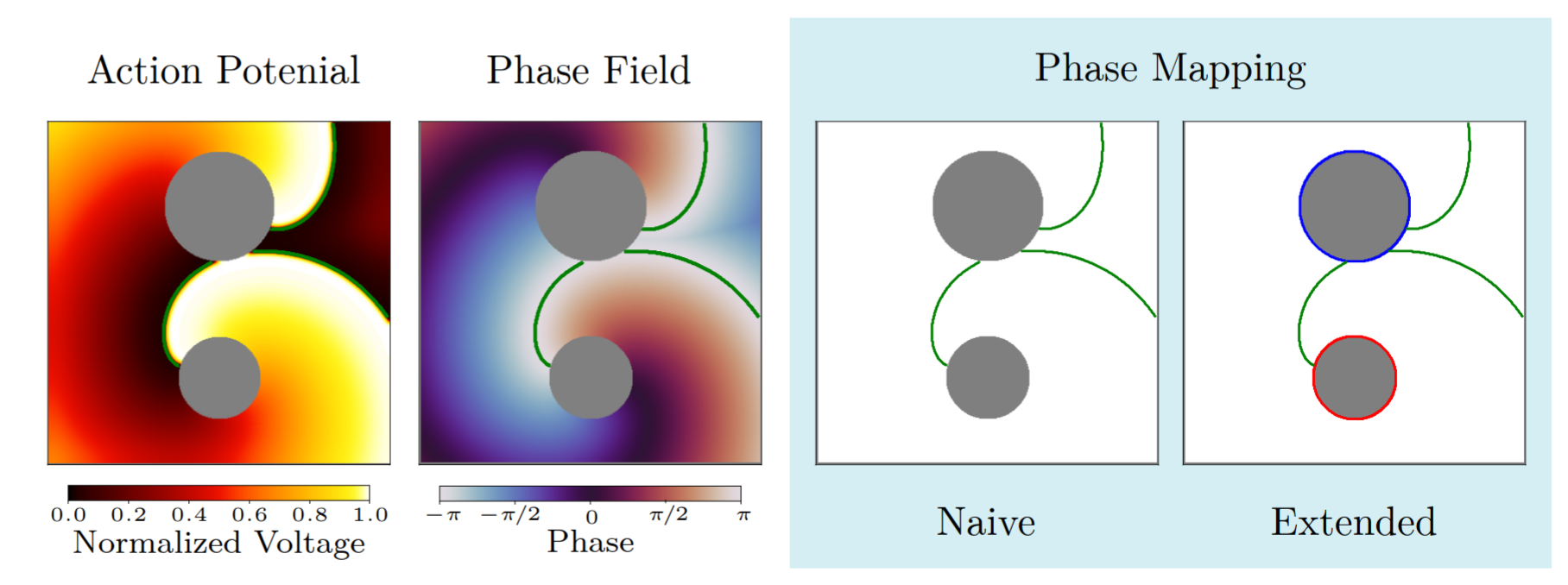 The naïve approach detects no phase singularities, as the phase within scar tissue is undefined. The extended approach instead identifies an average of two critical phase defects per time frame, with indices of +1 and −1 respectively.
The naïve approach detects no phase singularities, as the phase within scar tissue is undefined. The extended approach instead identifies an average of two critical phase defects per time frame, with indices of +1 and −1 respectively.Topology
OpenDGM also has algorithms that compute the index of given boundaries, for more on this see DGM-TOP